Quick summary: The human microbiome matters, but not every claim made about it deserves the same level of confidence. Some findings are solid, some are promising but still early, and some popular ideas move much faster than the evidence. To understand the microbiome properly, you have to separate what is biologically plausible from what is actually demonstrated, and what is clinically exciting from what is clinically real.
Editorial note: This article is intended for educational purposes only. It does not provide medical diagnosis, treatment, or personal health advice, and it should not replace consultation with a qualified healthcare professional.
Few ideas in modern biology have caught on so quickly while remaining so unevenly understood as the human microbiome.
It now shows up everywhere: in conversations about digestion, immunity, metabolism, inflammation, mood, aging, and disease. That popularity reflects something real. The microbes that live in and on the body are not passive passengers. They participate in processes that matter.
But the public conversation often outruns the science. A study finds that people with a certain condition have a different gut microbiome, and the result is treated as an explanation. A company offers a gut test, and the marketing implies it can tell you how to eat, how to live, and what is wrong with your body. A probiotic changes the microbial profile in a stool sample, and suddenly it is framed as a meaningful health solution.
That is where the subject gets genuinely interesting. The serious question is not whether the microbiome matters. It does. The serious question is how it matters, how much it matters, under which conditions, and with what level of evidence.
That distinction changes everything. Important is not the same as understood. And biologically interesting is not the same as clinically proven.
Contents
What is the human microbiome, exactly?
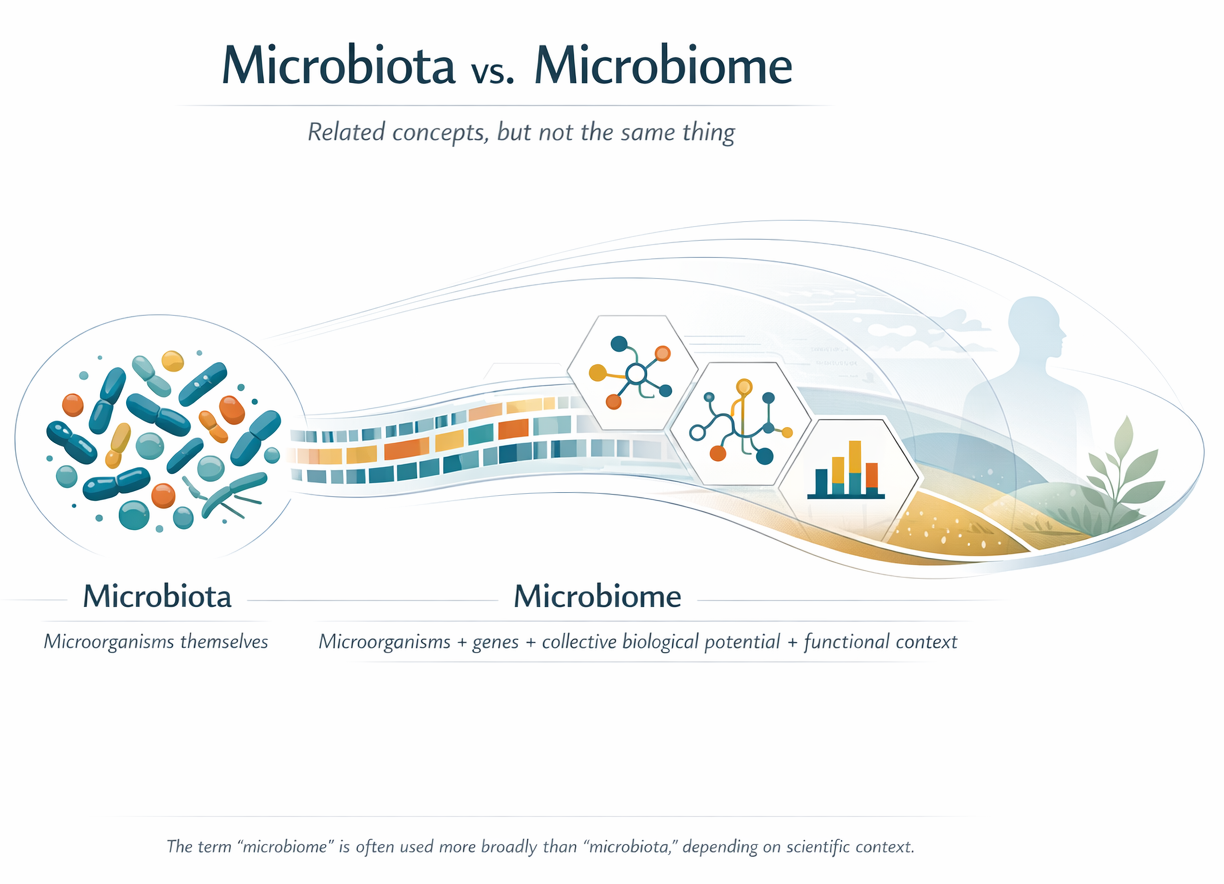
The first useful distinction is a simple one. Microbiota usually refers to the microorganisms themselves: the bacteria, archaea, fungi, viruses, and other microbes living in a particular environment. Microbiome is often used more broadly. Depending on context, it can refer to those organisms plus their genes, their collective biological potential, and sometimes even the surrounding environment in which they function.
In everyday writing, the distinction between the two often gets blurred. That is understandable, but it matters because the field is already complicated enough without muddy language. If you want to understand microbiome science clearly, it helps to know whether someone is talking about the organisms, their genes, or the broader biological system.
Most public discussion focuses on the gut, and not without reason. The gut contains a dense microbial community, it is constantly exposed to food, and it sits next to a large and dynamic immune interface. But the human microbiome is not just the gut. The skin, mouth, airways, and urogenital tract also host distinct microbial ecosystems, each shaped by very different physical and chemical conditions. As Figure 1 makes clear, “the human microbiome” is distributed across multiple body sites rather than concentrated in a single microbial compartment.
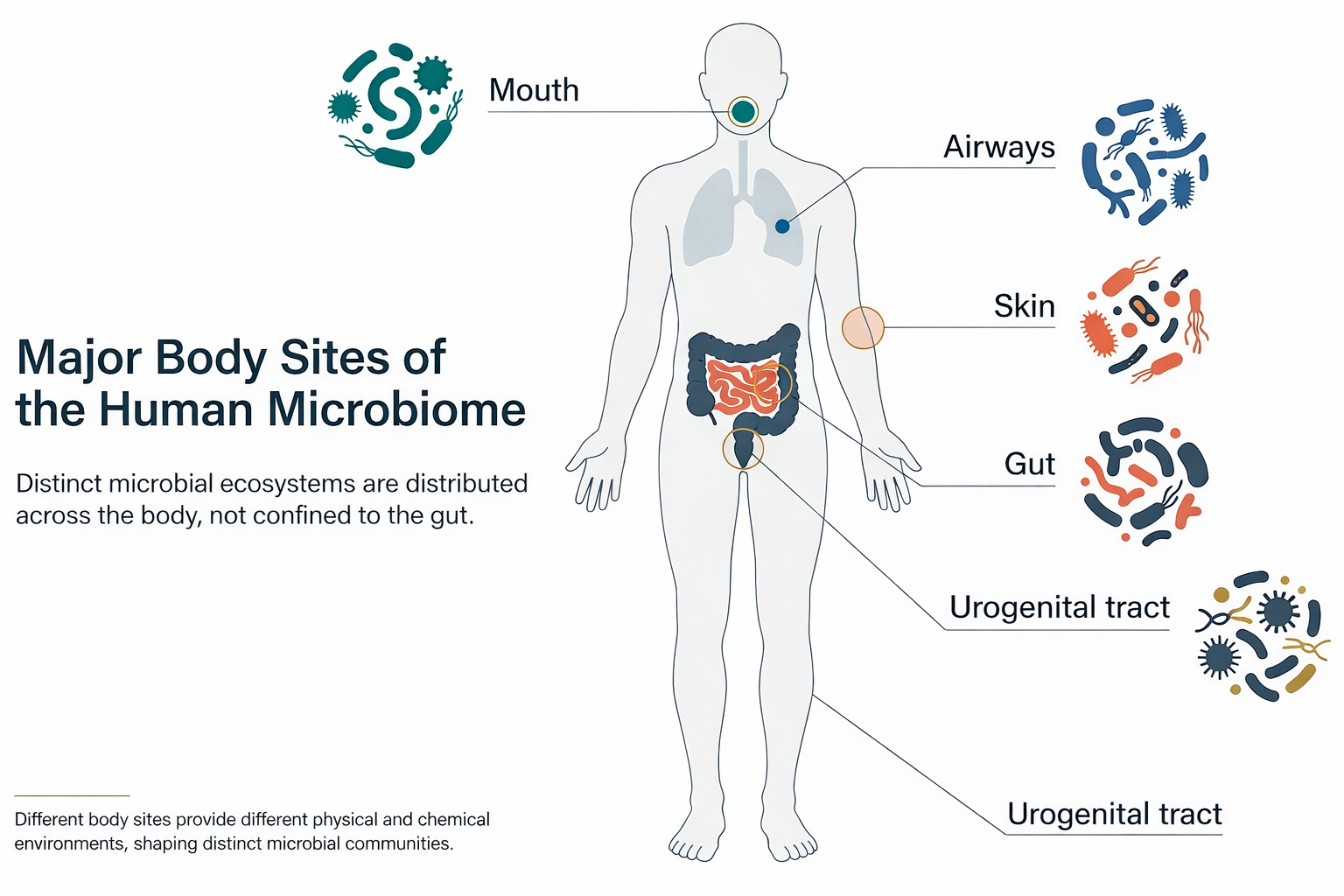
Even the phrase “the human microbiome” can be misleading if taken too literally. It sounds singular and stable, almost like an organ with a standard blueprint. It is neither. A better way to think about it is as a set of microbial ecosystems that vary from place to place and person to person.
That distinction between microbiota and microbiome also becomes easier to hold in mind when you see them side by side: one points to the organisms themselves, while the other points to a broader biological system that includes those organisms, their genes, and their functional context.
Variation is built into the biology
This point is easy to underestimate. Many people encounter microbiome science through the idea that there is some ideal community of microbes we are all supposed to have. But healthy people can differ from one another quite dramatically.
Two people can both be healthy and still have different microbial profiles because they eat differently, live in different environments, use different medications, have different life histories, or simply host different stable microbial communities. One person may eat a fiber-rich diet, another may not. One may have taken antibiotics twice that year, another not at all. One may travel often, another barely leave home. Those differences are not incidental to the microbiome. They help shape it. Figure 3 captures this point well: different microbiome configurations can still sit comfortably within a healthy biological range.
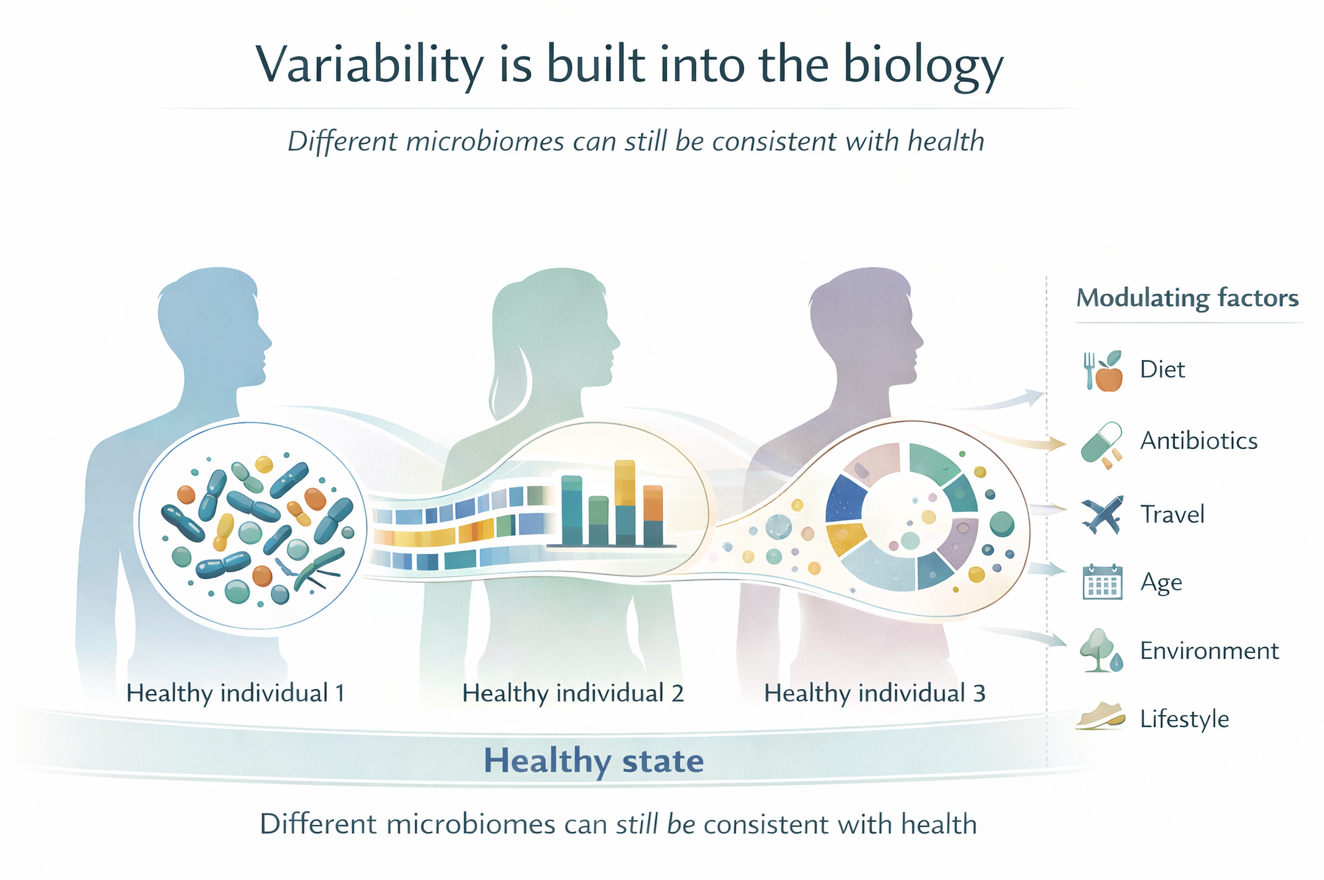
Even within one person, the microbiome can shift over time with diet, illness, antibiotics, travel, age, inflammation, and other exposures.
That variability is not noise around the edges. It is one of the field’s central biological facts.
Why does the microbiome matter for human health?
The microbiome became so influential in biology because researchers realized it was not just a catalog of organisms. It was a source of biological activity.
Gut microbes interact with food components that human enzymes do not fully process on their own. They transform part of what we eat into other molecules, including compounds that can affect the cells lining the gut, influence immune pathways, and shape aspects of metabolism. In other words, the microbiome is not just present in the gut. It helps create part of the gut’s chemical environment.
One useful way to picture this is to think of the microbiome as a living metabolic layer between the body and the outside world. Food enters. Microbes act on part of it. Some of the resulting products then become part of the conversation between diet, gut tissue, immunity, and physiology. Figure 4 gives that logic a visual form: environmental inputs are transformed by gut microbes into biologically active outputs that matter to host biology.
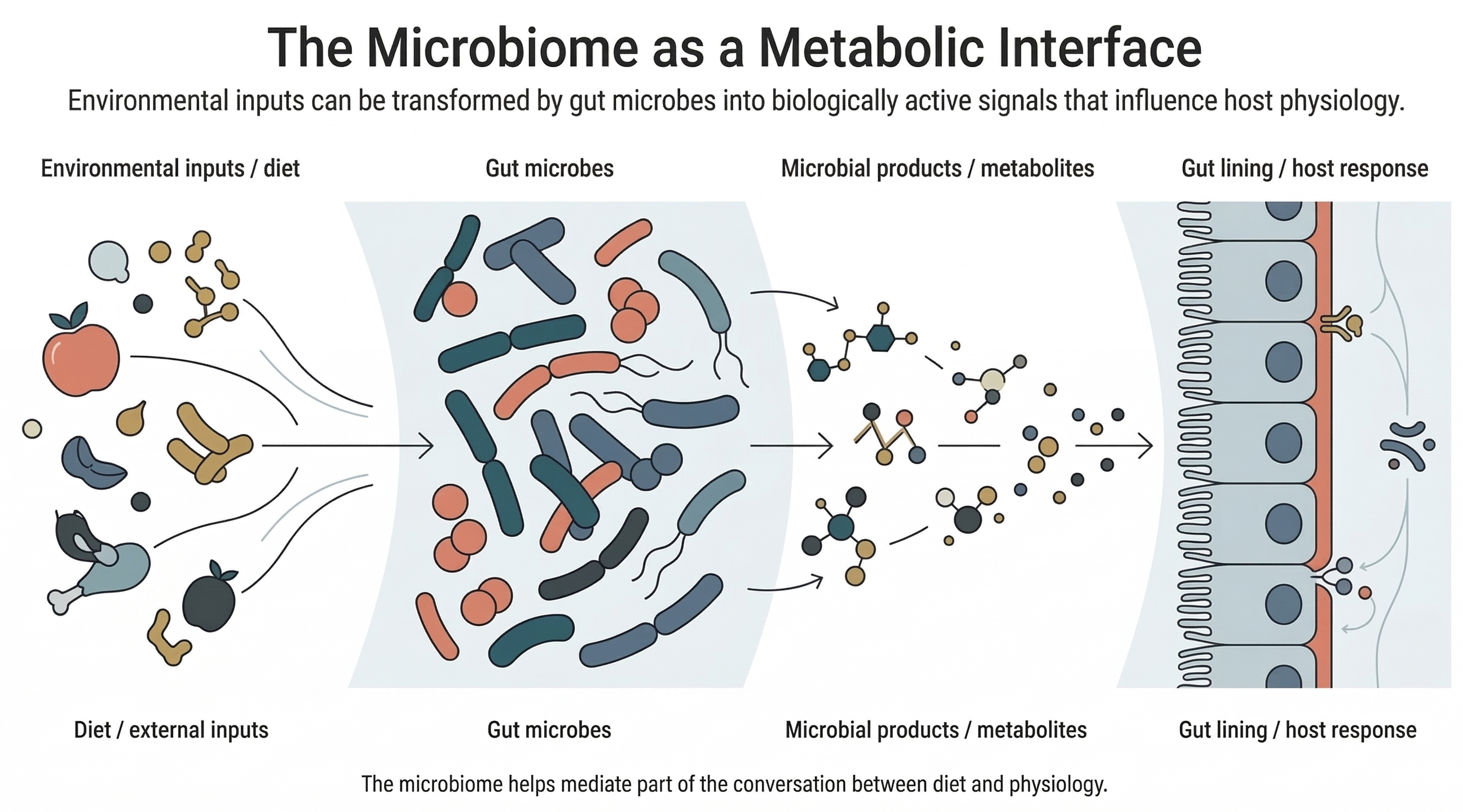
That is one reason the field captured so much attention. Once it became clear that microbes could generate biologically active molecules, it suddenly seemed possible that they might be involved in far more than digestion alone.
But this is where the field requires discipline. A mechanism that sounds plausible is not automatically a mechanism that has been shown to matter in real human disease. Biology is full of processes that can happen under certain conditions without turning out to explain much about broad health claims.
A plausible pathway is a starting point. It is not a final answer.
Why association is not the same as causation
This is the most important distinction in the whole field.
Many studies show that people with a certain disease or physiological condition have a different microbiome from people who do not. That may be important. But it does not automatically mean the microbiome caused the condition.
There are at least three broad possibilities. The microbiome may have helped drive the condition. The condition may have altered the body, and that may have changed the microbiome. Or both may be shaped by other factors at the same time. Figure 5 is useful here because it lets you see, in one glance, why a microbiome difference cannot be treated as a causal explanation by default.
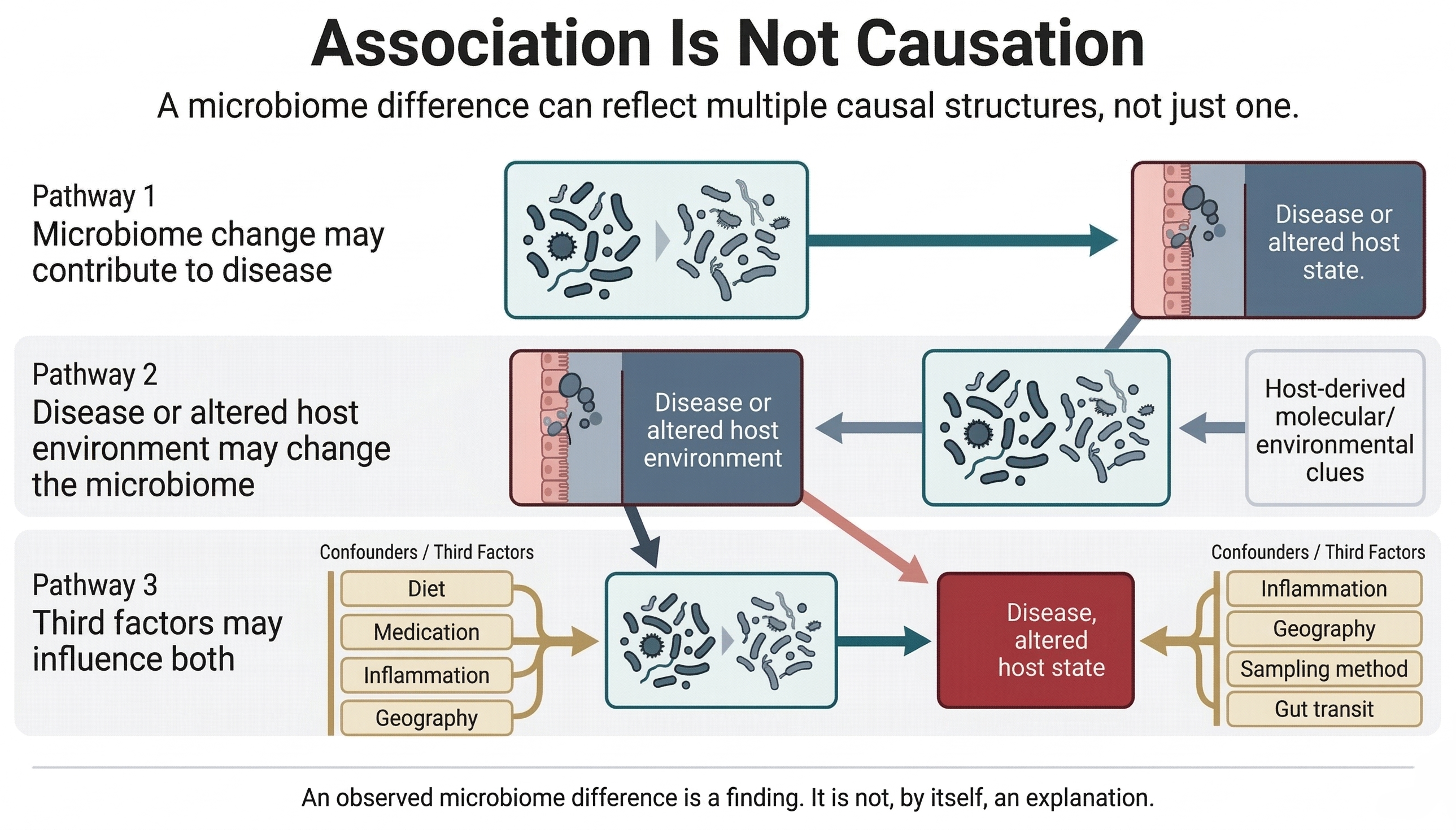
That second possibility is especially important and often overlooked. This is what scientists mean by reverse causality. An inflamed gut, for example, is not the same environment as a healthy one. Changes in oxygen levels, mucus, immune activity, motility, and nutrient availability can all alter which microbes thrive there. In that case, the disease is not simply the result of a microbial shift. The microbial shift may be one of the consequences of disease.
Common confounders in microbiome research
The picture becomes even more complicated because many other factors can distort interpretation. These are often called confounders, which is simply a technical way of saying hidden influences that can make an apparent relationship look more direct than it really is.
Some of the most common include:
- diet
- antibiotics and other medications
- proton pump inhibitors and laxatives
- age
- geography and lifestyle
- how quickly material moves through the gut
- inflammation itself
- differences in sampling and analysis methods
A very ordinary example helps. Imagine someone gets an infection, takes antibiotics, eats differently for a week because they feel unwell, and then develops gut symptoms. If you later find a changed microbiome, what exactly caused what? The answer may not be simple at all.
A changed microbiome is a finding. It is not, by itself, an answer.
This is one reason microbiome studies can be hard to reproduce. One cohort may show a strong pattern, while another does not, because the biological background, the environment, the medication profile, the diet, or the analytical method differs.
The microbiome field does not suffer from a lack of associations. It suffers from the much harder problem of showing which associations truly matter.
What we probably overstate about the microbiome
The first thing we probably overstate is the idea that there is one universally healthy microbiome. Current evidence does not support that. Healthy microbiomes can differ substantially across individuals. There may be broad features associated with health in some contexts, but the idea of one perfect microbial template is too simple for the biology.
We probably also overstate the meaning of diversity. You will often hear that more microbial diversity is always better. Sometimes diversity does correlate with desirable features. But not always, and not in a way that justifies treating diversity like a universal score of wellness. A community is not automatically beneficial simply because it is more varied.
The term dysbiosis is another problem area. It can be useful as a broad description of microbial disruption, but it is often used so loosely that it starts to sound like an explanation for almost anything. If someone has symptoms and their microbiome differs from a reference group, that difference is often labeled dysbiosis. But a label is not the same as a mechanism, and it is certainly not the same as proof of cause.
Commercial microbiome tests are also widely overstated. They often imply a level of interpretive precision that current science does not support. The field is still working through basic questions of reproducibility, standardization, functional interpretation, and clinically meaningful thresholds. A stool profile may reveal interesting information, but the leap from microbial readout to confident personal recommendation is often much larger than the marketing suggests.
Probiotics are frequently overstated for similar reasons. The word probiotic sounds singular, but it actually refers to a wide range of strains, doses, formulations, and clinical contexts. They are not interchangeable. Asking whether probiotics work is a bit like asking whether drugs work. Which one? For what condition? At what dose? Measured against which outcome?
Changing the microbiome is not automatically the same as improving health. Figure 6 organizes several of these recurring overstatements against what the evidence more cautiously supports, which is often a more qualified and more scientifically useful position.

What science supports more confidently
After all that caution, it is worth being equally clear about what stands on firmer ground.
We have strong reason to say that the microbiome is biologically relevant to human life. More specifically, we know that microbial communities:
- participate in digestion and nutrient transformation
- produce biologically active molecules
- interact with the cells that line body surfaces, especially the gut
- influence aspects of immune development and immune regulation
That is not trivial. It means the body does not operate in isolation from its resident microbes. Host biology is shaped through interaction.
What needs to be held carefully is the scale of the claim. There is stronger evidence for the microbiome’s general biological importance than for many of the specific public claims made in its name. That distinction matters because it lets us say something strong without saying something careless.
Yes, the microbiome matters. No, that does not mean it explains every chronic symptom, every disease pattern, or every wellness trend currently being attributed to it.
Where microbiome medicine is already clinically real
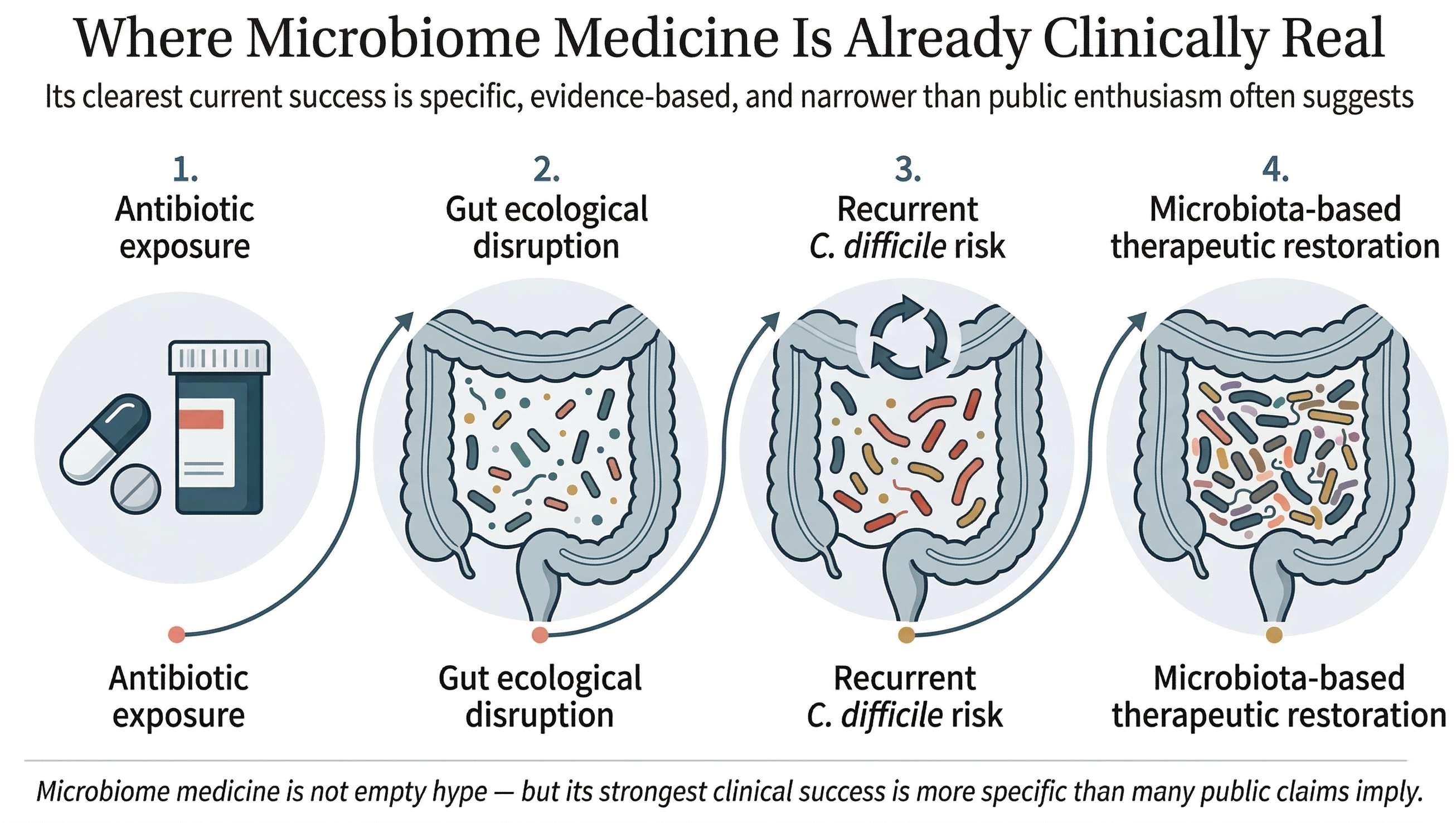
A balanced article on the microbiome should not end in blanket skepticism, because that would be misleading too. The field has already produced a real clinical success story, and it is an important one: recurrent Clostridioides difficile infection.
This case matters because it shows microbiome-based medicine can work when the clinical problem is specific enough and the biological link is strong enough. Recurrent C. difficile infection often follows major disruption of the gut microbial ecosystem, especially after antibiotic exposure. In that context, restoring a healthier microbial community is not a vague wellness idea. It is a targeted therapeutic strategy.
A short course of antibiotics can be life-saving. It can also, in some cases, sharply disrupt gut ecology. That is one reason this disease became such an important proving ground for microbiome medicine: the ecological damage is not hypothetical. It can be dramatic.
That is why microbiota-based therapies for preventing recurrence of C. difficile have become one of the clearest real-world achievements of microbiome medicine. Figure 7 closes the clinical argument by showing that progression directly: antibiotic exposure, ecological disruption, recurrent risk, and targeted microbiota-based restoration.
Clinically real, but more limited than the hype
This is the right way to hold the field in mind. Microbiome medicine is not empty hype. But its strongest successes are, at least for now, much narrower and more specific than the public story often suggests.
Recurrent C. difficile matters so much because it shows that microbiome medicine can be genuinely clinical, while also reminding us how much narrower real success is than public enthusiasm usually suggests.
That is not disappointing. It is what serious science looks like when it starts becoming medicine.
Why so many microbiome interventions disappoint
One of the simplest and most useful sentences in this whole subject is this:
Changing the microbiome is easier than proving that the change matters.
An intervention may change microbial composition without improving symptoms, reducing risk, or altering the course of a disease in a meaningful way. A statistically significant shift in sequencing data is not automatically a clinically meaningful result.
Part of the problem is that composition alone does not tell the whole story. Knowing which organisms are present is useful, but it does not fully reveal what they are doing. Put differently, knowing which microbes are present is a bit like knowing who is in the room. Useful, yes, but not the same as knowing who is speaking, who is doing the work, and who is changing the course of the conversation.
Two people can host different microbial communities that perform similar functions. And two communities that look similar on paper may behave differently depending on diet, inflammation, host physiology, or other ecological conditions.
This is why the field has been moving away from simplistic lists of “good” and “bad” bacteria. Biology is usually more interesting than that, and less cooperative with those categories.
Who is there matters. But what they are doing, under which conditions, may matter even more.
What the next phase of microbiome science will require
The next phase of microbiome science will probably be less impressed by broad claims and more committed to sharper questions.
That means more longitudinal studies, which follow people over time rather than relying only on snapshots. It means better control of confounding factors. It means stronger methodological standardization, so that results from different studies can be compared more meaningfully. And it means deeper integration of microbial data with host biology, metabolite measurements, clinical outcomes, and mechanism.
This is where the field is heading: away from fascination with microbial lists and toward a more integrated biology of function, mechanism, and context.
That shift is healthy. It replaces a shallow question — does the microbiome matter? — with better ones:
- Which microbial functions matter?
- In which people?
- In which tissues?
- Over what timescale?
- By what mechanism?
- And with what measurable consequence?
That is slower science than hype would prefer. But it is better science.
The microbiome deserves attention not because it explains everything, but because it pushes biology to think more carefully about interaction, ecology, and evidence. Its future will not be built by bigger claims. It will be built by better questions.
Frequently asked questions
What is the human microbiome?
The human microbiome is the broader biological system formed by the microorganisms that live in and on the body, along with their genes and, depending on context, their functional environment. In everyday usage, people often use it more loosely to mean the body’s resident microbes, especially those in the gut.
What is the difference between microbiota and microbiome?
Microbiota refers to the microorganisms themselves. Microbiome is broader and may include the organisms, their genes, and their collective biological potential. The distinction is not always used perfectly consistently, but it is still useful.
Can the microbiome really cause disease?
Sometimes it may contribute to disease, but that is much harder to prove than many headlines imply. In many cases, microbiome changes may reflect disease, treatment, inflammation, or other background factors rather than acting as the primary cause.
Why do healthy people have different microbiomes?
Because the microbiome is shaped by diet, environment, age, medication use, life history, and many other influences. Different microbiomes are not necessarily a sign of dysfunction. Variation is part of the biology.
What does dysbiosis actually mean?
At its most basic, dysbiosis means a disrupted or altered microbial community. The problem is that the term is often used too loosely. It can describe a difference without proving what that difference means, how it arose, or whether it is clinically important.
Do probiotics really work?
Sometimes, in specific contexts. But probiotics are not one thing. Different strains can have different effects, and evidence depends on the exact product, dose, condition, and outcome being studied. Broad claims about probiotics are usually much less reliable than condition-specific ones.
Are gut microbiome tests actually useful?
For most people, not in the precise and personalized way they are often advertised. They may be interesting, but current science still does not support the kind of confident individual guidance many commercial services imply.
Is there one ideal healthy microbiome?
No. Healthy people can have quite different microbiomes. There may be patterns associated with health, but current science does not support the idea of one universal ideal community that everyone should match.
Suggested reading
If you want to keep exploring related ideas in GlobalBioverse, these articles connect especially well with broader questions about biology, environment, and interpretation:
Explore more in GlobalBioverse
Visit the GlobalBioverse hub to explore more English-language Biology content.
Go to GlobalBioverseSupport this project
If this article helped you understand the topic more clearly, consider supporting this project. Your contribution helps sustain independent, rigorous, and accessible Biology education for students, educators, and curious readers.
Support this project